Enhanced TDS
Identification & Functionality
- Chemical Name
- Molecular formula
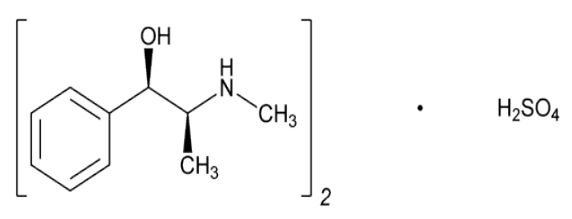
- (C₁₀H₁₅NO)₂ · H₂SO₄
- Technologies
- Product Families
- Definition
Ephedrine Sulfate contains not less than 98.0 percent and not more than 101.0 percent of (C₁₀H₁₅NO)₂ · H₂SO₄, calculated on the dried basis.
- Chemical Structure
Applications & Uses
Properties
- Physical Form
- Soluble In
- Typical Properties
Value Units Test Method / Conditions Molecular Weight 428.54
Regulatory & Compliance
- Certifications & Compliance
- Chemical Inventories
- FDA Disclaimer
These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
- USP Reference Standards
USP Ephedrine Sulfate RS
Storage & Handling
- Storage Information
Preserve in well-closed, light-resistant containers.
