Enhanced TDS
Identification & Functionality
- Chemical Name
- Pharma & Nutraceuticals Functions
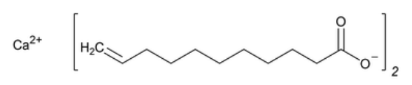
- Molecular formula
- C₂₂H₃₈O₄Ca
- Technologies
- Product Families
- Definition
Calcium Undecylenate contains not less than 98.0% and not more than 102.0% of calcium undecylenate (C₂₂H₃₈O₄Ca), calculated on the dried basis.
- Chemical Structure
Applications & Uses
Properties
- Physical Form
- Appearance
- white powder
- Soluble in
- Water, Chloroform, Acetone, Ether, Cold Alcohol
- Typical Properties
Value Units Test Method / Conditions Molecular Weight 406.62 - - - Specifications
Value Units Test Method / Conditions Melting Point 66 - 67.5 °C Assay (on dried basis) 98.0 - 102.0 % Loss on Drying 2.0 - 5.7 % Particle Size (Passes through a No.100 seives) min. 99.0 % Limit of Free Undecylenic Acid max. 0.1 %
Regulatory & Compliance
- Certifications & Compliance
- Chemical Inventories
- FDA Disclaimer
These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
- USP Reference Standards
- USP Calcium Undecylenate RS USP Undecylenic Acid RS
Storage & Handling
- Shelf Life
- 2 Years
