Enhanced TDS
Identification & Functionality
- Chemical Name
- Molecular formula
- C₈H₁₀N₄O₂ · H₂O
- Technologies
- Product Families
- Definition
Caffeine is anhydrous or contains one molecule of water of hydration. It contains not less than 98.5% and not more than 101.0% of C₈H₁₀N₄O₂, calculated on the anhydrous basis.
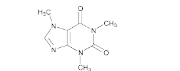
- Chemical Structure
Applications & Uses
Properties
- Physical Form
- Soluble In
- Appearance
- White crystalline powder
- Typical Properties
Value Units Test Method / Conditions Melting Point 236.6 °C - Molecular Weight 194.19 Acidity (0.01 mol/L NaOH) max. 0.2 ml - Organic Impurities (individual impurity) 0.02 % - Total Impurities 0.03 % - Sulfate Content max. 500 ppm - Heavy Metals (as Lead) max. 0.001 % - Loss on Drying 0.05 % - Residue on Ignition 0.01 % - Sulphated Ash Content 0.01 % - Assay Content (on dried basis) 100 % HPLC Particle Size (Through 80 mesh) 90.2 % -
Regulatory & Compliance
- Certifications & Compliance
- Chemical Inventories
- FDA Disclaimer
These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
- USP Reference Standards
- USP Caffeine RS
Packaging & Availability
- Labelling Information
Label it to indicate whether it is anhydrous or hydrous.
